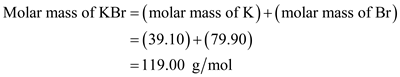
Without knowing the chemical or molecular formula of the compound and without knowing the atomic weights of the individual elements involved in the chemical formula it is very difficult to calculate the molar mass of the respective compound. Potassium is a chemical element with the symbol K. and your answer to the following textbook question: To two decimal places, what is the relative atomic mass and the molar mass of the element potassium, K.

The molar mass of the potassium permanganate is 158 g/mol. Now we must express the molecular weight of the potassium permanganate in molar mass.
POTASSIUM MOLAR MASS HOW TO
The chemical formula of the potassium permanganate is $KMn$ = 19 + 55 + 4 (16) = 158. Explanation of how to find the molar mass of K3PO4: Potassium phosphate.A few things to consider when finding the molar mass for K3PO4:- make sure you have t. Molar mass of potassium is 39.09830 0.00010 g/mol Compound name is potassium Get control of 2022 Track your food intake, exercise, sleep and meditation for free. We should know the chemical formula of the potassium permanganate to calculate the molar mass of it. Calculate the mass of potassium chlorate required to liberate 6.72 dm of oxygen at STP. In the question it is asked to calculate the molar mass of the potassium permanganate. We should know the molecular or chemical formula of the respective compound.
Hint: The molar mass of any compound can be calculated by adding the atomic weight of the individual atoms involved in the chemical formula of the respective chemical compound.
